With its main combustion product being water vapor, hydrogen fuel is the holy grail of clean combustion for electric power generation. The downside? Hydrogen gas is not readily available and must be produced.
In this article, we look at a technology that can generate hydrogen and power from gasification of a blend of biomass and coal with a “net negative” CO2 footprint. A recent US DOE-sponsored study concludes that:
- Coal gasification (plus biomass) enables a beneficial switch from natural gas feedstock for blue (low carbon) H2 production, which
- Significantly reduces the carbon footprint of hydrogen production (by almost 80%) while increasing the electricity generation (by 60%), and
- With legislated tax credits and other financial incentives, the economics can work out favorably as well
- The switch can turn coal into a useful tool of decarbonization instead of letting it go unused underground.
If hydrogen production, storage, and transportation are CO2-free, then the mission of CO2-free power generation is accomplished. This is potentially (but not necessarily and not fully) true for H2 production via electrolysis of water using CO2-free power, e.g., wind, solar, hydro, or nuclear. This is called “green” hydrogen (or “pink” in the case of nuclear).
While these technologies generate power without CO2 emissions, their “greenness” is not automatically given. Only excess kilowatts from these resources can be considered green. Otherwise, each kilowatt diverted from the electric grid to electrolysis of water to make H2 is replaced by a kilowatt with a certain CO2 footprint.
Due to the limited availability of such “surplus” green energy and the exorbitant power consumption of the electrolyzers (roughly 50 kWh/kg H2 produced), there is a strict limit to the availability of truly “green” H2 , which, by the way – at the time of writing and at least in the near term – is too expensive to make economic sense.
Hydrogen produced using steam methane reforming (SMR), the established thermal technology to make economical hydrogen, comes with a substantial CO2 footprint, i.e., 9-10 kg of CO2 emitted for each kg of H2 produced (plant stack basis, not life cycle).
The redeeming feature of SMR-produced H2 is its low cost. The remedy proposed to alleviate its heavy CO2 baggage, “blue” H2, adds carbon capture to the conventional SMR plant. This may be an attractive candidate for industrial H2 production without concomitant CO2 emissions.
However, for power generation purposes, this involves a trade-off because the most commonly used SMR feedstock, natural gas, can be used directly in a GTCC power plant with the lowest possible CO2 footprint of all the fossil fuel-based technologies (60-70% less than that of coal-fired power plants).
By adding post-combustion CO2 capture, it can be nearly CO2-free as well. Each kilogram of blue H2 using natural gas as feedstock has an “opportunity cost” in missed CO2-free electricity generation, which is substituted by the grid with its own CO2 footprint.
Gasification for Blue H2
Luckily, SMR (and its up-and-coming alternative, autothermal reforming {ATR}) – more on that below – is not the only thermal technology to make blue H2 from a hydrocarbon feedstock.
Gasification of coal or other hydrocarbon feedstocks is used to produce a syngas, which, after being scrubbed of CO2 (along with H2S for sulfur removal), can be fed to an adsorption process to separate high-purity H2 from the remaining components. And, as in any other carbon capture process, the captured CO2 can be sequestered or utilized in an industrial process such as enhanced oil recovery.
Compared to SMR, blue H2 from coal is more expensive – but, at least for now, still less expensive than green H2 produced via electrolysis of water. Any in-depth cost comparison depends strongly on respective feedstock prices and the gasification technology used. Other factors that will play a role in comparative ranking of H2 production options are government policies such as tax credits, future advances in electrolyzer technology, and increased deployment lowering unit prices.
The underlying attractiveness of blue H2 from coal (or other environmentally problematic hydrocarbon feedstocks) stems from it being one potential way to make a “green” product from an otherwise unusable – especially in North America and Europe – but ample energy resource.
That way, natural gas can be reserved for low-carbon electric power generation in gas turbine power plants to replace coal use. By blending coal with carbon neutral biomass (as per US EPA guidelines), it is even possible to achieve “net negative” carbon emissions as well.
Coal+biomass = “net negative” H2
In a recent study funded by the U.S. DOE, a conceptual design has been developed for a pilot plant to produce net negative carbon H2 and power from coal gasification using biomass. A simplified block diagram of the plant is provided in Figure 4.
The process feedstock is Powder River Basin (PRB) subbituminous coal and corn stovers in a 50/50 composition (dry weight basis). Raw syngas, primarily CO + H2, is produced in an oxygen-fed, pressurized gasifier and scrubbed in cyclone separators to remove particulate recycled in the gasifier. Syngas treatment includes water-gas shift (converts the CO to CO2 plus additional H2 by reacting with H2O), gas cooling (generates HP steam), and acid gas (CO2 and H2S) removal via chemical absorption.
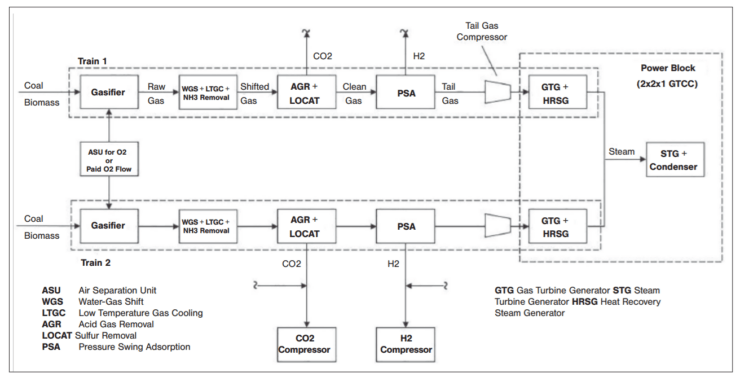
Treated, desulfurized hydrogen-rich syngas then undergoes pressure swing adsorption to separate 99% pure H2 from the gas stream. The remaining “tail” gas (mostly CO + H2) is burned in a gas turbine combined cycle power plant to generate electric power. Part of the generated power is utilized within the plant and the balance is fed to the grid.
The GTCC power plant
The tail gas from the PSA unit, containing 34%(v) H2 is fed to the 2x2x1 GTCC power plant driven by two Siemens SGT-800 industrial gas turbines (Figure 5). According to Siemens Energy, the DLE combustion system of the SGT-800 gas turbine can burn high-hydrogen fuel gas with low CO and NOx emissions, in compliance with the current US EPA regulation, without diluent injection.

The three-pressure (non-reheat) bottoming steam cycle is integrated with the coal gasification island and syngas process blocks. Carbon dioxide (produced from combustion of CO in tail gas) exiting the HRSG stack gas is not captured. Power block gross output is 134 MWe and gross thermal efficiency of the power block is 56.7% (LHV). However, 84 MWe is consumed by gasification plant systems, including the ASU (~30 MW) and the CO2 compressor (~18 MWe).
Thus, a net power plant output 50 MWe is supplied to the grid. (In the unique case of oxygen available from a nearby industrial facility for over-the-fence supply, output to the grid increases to ~80 MWe.)
Co-gasification vs alternatives
Looking at thermal performance of the gasification to H2-and-power co-generation facility, feedstock consumption is, on an as-received basis, ~2,000 STPD coal and ~1,600 STPD corn stover (686.5 MWth HHV total). Overall cogeneration efficiency, based on hydrogen production of 8,500 kg/h (283.3 MWth LHV) and net power output (50 MWe), is 48.7% HHV (52.9% HHV if oxygen is procured from over-the-fence supplier).
Assuming an optimistic 60% conversion efficiency for the hydrogen product (i.e., assuming that the power plant burning the H2 is next to the gasification/cogen plant and no storage, transport, leakage, and other performance debits), effective electric efficiency is 32% HHV (36.4% HHV if oxygen is procured from outside the plant battery limit).
For comparison to SMR, using an SMR thermal efficiency range of 65% to 75% on LHV basis (i.e., LHV of hydrogen produced as a fraction of the LHV of natural gas feedstock), with the same optimistic 60% conversion efficiency (with the caveats already mentioned), the effective electric efficiency burning grey H2 via SMR is 0.65 to 0.75 x 0.6 or 39% to 45% LHV.
There are two options to make the product chain from natural gas feedstock to power plant generator terminals (almost) carbon neutral: (1) pre-combustion: producing “blue” H2 by adding carbon capture to the SMR; or (2) post-combustion: adding CO2 capture to the GTCC.
The penalty would be from 10% to 15% depending on a multitude of factors. Thus the efficiency range would be around 33% to 40% LHV, which, on HHV basis, corresponds to 30% to 36%.
So, on a performance-only basis, the net negative carbon coal + biomass gasification for H2 and power cogeneration readily competes with blue H2 via SMR. Actually, it is significantly better than just “competitive”. This is due to (1) net-negative carbon footprint – impossible to match by SMR – and (2) significant losses and/or inefficiencies in transit between the SMR plant and the power plant using the blue H2 produced. Thus, the cost comparison between the two technologies becomes the key differentiating factor.
Cost comparison – it all depends
Steam reforming of natural gas is a mature technology widely used for industrial hydrogen production. It is the least expensive, at about $1/kg, or less, as indicated in the following report (last updated in June 2019).
With carbon capture, the price is predicted to end up from around $1.5/kg in the USA to more than $2/kg in Europe (the same source). This is a tough benchmark to beat.
In addition to CAPEX and OPEX of the co-gasification plant (a Phase 2 FEED study for Class 3 estimate is in the works), the levelized cost of H2 (LCOH) depends on the cost of the corn stover pellets (significantly more expensive than coal), sale price of electricity, and financing costs. The location of the plant plays a significant role too (e.g., access to coal and corn stover as well as CO2 sequestration sites).
Phase I study calculations showed that with Inflation Reduction Act (IRA) 45Q credits, and corn stover pellets costing about the same as PRB coal, an LCOH of $2/kg or less is possible. (At the time of the study economic analysis, the 45V legislation was not yet released.)
Why coal (and/or biomass)?
Hydrogen is an important industrial feedstock. Current H2 production is primarily via chemical/thermal means, to an overwhelming extent by steam reforming of natural gas (mostly methane) with a significant CO2 footprint. A transition to H2 production technologies with little or no CO2 emissions is a critical step towards slowing down and, ultimately, reversing the global warming caused by greenhouse gas (GHG) emissions.
Another significant step in that direction is decarbonization of the electricity generation sector. Replacement of coal with natural gas has been a strong positive factor in this endeavor (the record in the U.S. being a prime example as noted at the outset). Especially with advanced class gas turbine combined cycle (GTCC) power plants equipped with 90-95% post-combustion carbon capture, near carbon-free electricity generation efficiency can still be well above 50% LHV.
Redirecting natural gas from electricity generation to hydrogen production beyond the existing levels used by industry is not a viable decarbonization move. If the blue hydrogen thus produced is used for electricity generation, the end result is increasing the CO2 emissions – just compare the efficiencies calculated above, i.e., low to mid-30s% LHV for SMR with carbon capture. If the end use of the blue hydrogen is industrial, it has an opportunity cost because it takes away gas-fired generation from possibly replacing coal-fired generation.
For all of its downside in terms of environmental unfriendliness, especially in the U.S., coal is a valuable energy resource. Once considered a panacea, “clean coal” for electric power generation has been tried and eventually dropped with practically zero possibility of coming back. “Clean coal” for hydrogen production is a potentially (but by no means guaranteed) cost-effective means to transform a plentiful and relatively inexpensive resource to valuable “blue” hydrogen.
Gas power to replace coal power
In evaluating the cost effectiveness of producing blue H2 via coal and/or biomass + coal gasification, one should also factor in the release of natural gas for replacement of coal-fired electricity generation with concomitant reductions in CO2 emissions.
A quick calculation can demonstrate the above assertion. In a roughly 75% efficient (LHV basis) steam reforming process, 3.26 kg CH4 (the main constituent of natural gas) is consumed to produce one kg of H2 with 10 kg of CO2 emissions.
At 60% net LHV efficiency, typical of a modern GTCC power plant, the opportunity cost of not using that CH4 in electricity generation is 27.2 kWh, with its 9 kg of CO2 emissions (i.e., 330 kg/MWh).
To generate the same electricity in a PRB subbituminous coal (7,500 Btu/lb LHV as received) fired SCPC thermal (steam) power plant with 40% net LHV efficiency, ~14 kg of coal is burned with 23.9 kg of CO2 emissions (880 kg/MWh).
Let us assume that the coal-fired plant is replaced by a CH4 burning GTCC and the same coal thus released is used in the coal-biomass fired H2-power co-production plant described above. Hydrogen production is 1.28 kg with negative 5.9 kg of CO2 emissions (as explained above). Note that to produce 1.28 kg of H2 via SMR, we need to consume 4.15 kg of CH4 with 12.8 kg CO2 emissions.
To summarize the results:
Base Case: 1 kg grey H2 from 3.26 kg CH4 via SMR plus 27.2 kWh electricity by burning 14 kg PRB coal in a thermal power plant.
Switch Case: 1.28 kg blue H2 plus 9.1 kWh electricity from 14 kg PRB coal (plus corn stover) in the coal gasification plant plus 27.2 kWh electricity in GTCC by burning 3.26 kg CH4 (not used in SMR for H2 production). The “switch” is two-fold:
• Hydrogen production feedstock switch from CH4 to coal (and biomass)
• Electricity generation fuel switch from coal to CH4
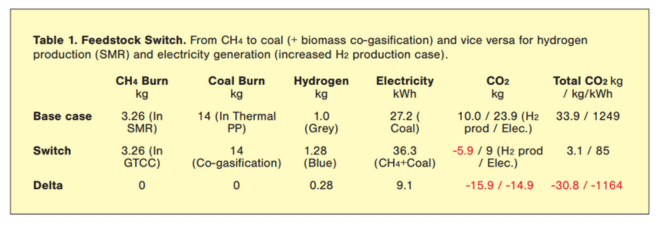
The comparison between the cases is summarized on Table 1. The “switch” increases both H2 production and electricity generation (each by one-third) while reducing CO2 emissions by 91% (mass basis, 93% in per MWh terms). Note that SMR electricity consumption, mainly H2 compression, is ignored.
An alternative comparison between cases is provided in Table 2, where H2 production via SMR (Base Case) is increased to match the higher H2 production rate via co-gasification (Switch Case). This increase in SMR H2 production enables additional fuel conversion, i.e., releases more CH4 from H2 production to electricity generation in a GTCC.

As shown, the result is even higher electricity generation (by nearly 60%) with 78% reduction in CO2 emissions by mass (86% in per MWh terms).
Autothermal reforming (ATR)
SMR is the dominant commercial technology for hydrogen production, but it is not the only reforming technology available. The up-and-coming autothermal reforming (ATR) is the other thermal technology of interest to produce hydrogen from natural gas as feedstock at commercial scale. And it has advantages over SMR that need to be considered when comparison is made to co-gasification plus gas treatment as a means of hydrogen production.
ATR combines partial oxidation (POX) and steam reforming of natural gas to produce a syngas (primarily H2+CO) with the inherent advantage of requiring no external heat input since the POX section is exothermic. As in co-gasification, hydrogen is separated from the shifted syngas in a PSA system, and byproduct CO2 may be captured. The disadvantage of ATR is that the POX section requires pure oxygen (to avoid introduction of nitrogen if air-blown), which is supplied by an external cryogenic ASU.
On average, ATR consumes 3.5 kg of natural gas per kg of H2 produced and it also comes with a substantial on-site CO2 footprint, i.e., 8.3-9.3 kg of CO2 emitted for each kg of H2 produced (with no capture). Compared to SMR this process benefits capturing the majority of CO2 (as much as 95% or higher) from the shifted syngas in a single (pre-combustion) stage.
For the same capture efficiency in an SMR plant, two steps are required: a post-combustion capture block (from the reactor flue gas) and a second pre-combustion capture block (from the shifted syngas). While ATR and SMR as blue H2 production technologies are readily comparable in performance, most published studies point to the ATR as the more cost-effective option.
As indicated, ATR combines POX with SMR. POX of natural gas by itself is not considered competitive with SMR, i.e., not a likely for future blue H2 production (see the article cited earlier for its performance [1]).
Methane pyrolysis or “cracking”
Another technology for blue H2 production from natural gas that must be mentioned for completeness is methane pyrolysis (also called methane cracking or decomposition), whose end byproduct of value is solid carbon or “carbon black”. (Hydrogen produced via pyrolysis is sometimes called “turquoise” rather than “blue” H2.)
This form of byproduct (vs. CO2 gas) clearly has significant advantages vis-à-vis CO2 capture via more complex amine-based chemical absorption, and sequestration. However, to bring methane decomposition temperatures down to a level comparable to SMR, a catalyst is required. (A plant using Monolith’s high temperature plasma torch and electricity to convert natural gas and nitrogen to carbon black with an ammonia byproduct, 14,000 tons/year, has been operating in Hallam, Nebraska since late 2020.)
To date, from a technology readiness perspective, catalytic methane pyrolysis is behind ATR – and likely to be costlier.
SMR vs. ATR vs. Co-gasification
Thus the technology competition for commercial blue H2 production from natural gas (mostly methane) will most likely be between SMR and ATR with carbon capture and storage. Representative process parameters, including those of coal gasification using biomass, are summarized in Table 3. For grey H2 production (no CO2 capture), ATR performance is at par with SMR except for power consumption (mainly due to the ASU).
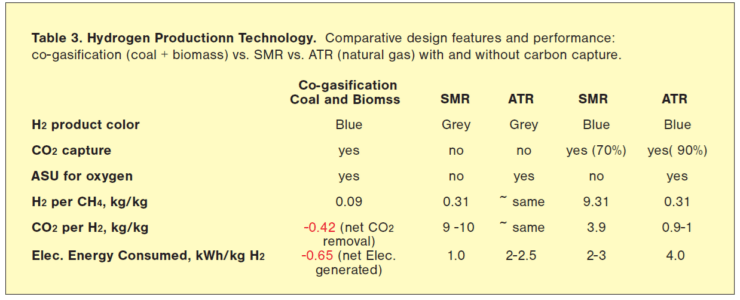
For blue H2 production (with CO2 capture), ATR is the better choice with a lower CO2 footprint than SMR. Several studies have evaluated the ATR pathway to be less expensive than SMR for blue hydrogen, especially at high capture efficiency from a single source, e.g., shifted syngas. (In SMR, 90% capture efficiency from shifted syngas only corresponds to about 70% effective CO2 capture.)
Looking at the recent trends, large scale ATR plants are expected to be a key producer of blue hydrogen and blue ammonia going forward. Thus, the feedstock switch scenario above (Tables 1 and 2) is repeated by replacing SMR (grey H2) with ATR+CCS (blue H2) in the base case (same H2 per CH4 ratio on mass basis). The results are summarized in Table 4.

Clearly, CO2 emissions benefit is less pronounced than the base case with SMR (no capture) but still significant plus with the additional improvement in the extra electricity column.
Since either technology is at an early stage of development, a reliable cost comparison at this stage would be subject to high uncertainty. An argument might be made for coal gasification in terms of technology maturity.
History, however, teaches us that each variation (presence of biomass) of a base technology brings its own challenges and requires careful design and development – with Kemper being the most recent example. Still, co-gasification of coal and biomass for H2 and electricity cogeneration is a worthy competitor in the blue hydrogen realm.
In conclusion…
Coal gasification using biomass makes possible a beneficial switch between two hydrocarbon feedstocks, which significantly reduces the carbon footprint for the same hydrogen production (by almost 80% on a mass basis) while increasing the electricity generation (by 60%).
With generous legislated tax credits and other financial incentives, the dollars and cents can work out favorably as well. This makes sense from the perspective of turning a valuable natural resource into a useful tool of decarbonization instead of letting it go unused underground.
Authors’ Note: The blue hydrogen production system described in this article is based on findings from a study supported by the U.S. Department of Energy under Award Number DE-FE0031993. In addition the co-author, Kiran Chaudhari, is now Engineering Study Manager, Energy Transition, Kiewit.
Manufacturing H2 using coal: a closer look at gasification and the gas treatment islands
The gasification technology used for the DOE study, GTI Energy’s U-GAS, is an advanced, single-stage, pressurized fluidized-bed process. Dry feedstock is fed by a lock hopper into the gasifier where it reacts with steam and oxygen (from an ASU) at 1,100 °C (~2,000 °F). The gasifier operating pressure is 30 bar.
Generated raw syngas (primarily CO and H2) is fed to dual cyclone separators to remove particulates that are recycled back into the gasifier. The hot syngas is cooled by generating high pressure superheated steam before passing through a filter system to remove fines, which are also recycled.
Water soluble contaminants, including ammonia, hydrogen chloride and hydrogen cyanide gases, are then removed in a wet scrubber and the scrubbed syngas gas is further processed in the Water Gas Shift (WGS) reactor to increase hydrogen content.
The WGS process (also referred to as sour gas shift) consists of reacting the gas mixture with steam at high temperature, resulting in “shifted” syngas, composed primarily of CO2 and H2 (about 47% and 42% by volume, respectively).
The shifted gas is fed to a Selexol Acid Gas Removal (AGR) unit to remove and recover H2S and CO2. This system is sized to remove CO2 generated from gasification of the coal plus a portion of the CO2 generated from gasification of the (carbon neutral) biomass; thus making the overall plant emissions net-negative carbon.
Of the total CO2 produced by the gasification process, about 60% is attributed to the coal with the balance attributed to the corn stover. Overall design capture efficiency is close to 70%.
A LOCAT unit is installed to treat the AGR off-gas for removal of remaining H2S. This aqueous-based catalytic process converts the H2S to elemental sulfur. Treating AGR off-gas containing only CO2 and H2S, rather than the full syngas stream, reduces equipment size and cost.
The primary constituents of the desulfurized clean syngas, H2
(about 75% by vol) and CO, are separated through a Pressure Swing Adsorption (PSA) process which delivers 99% purity hydrogen product. PSA comprises alternating steps of pressurization and depressurization driving adsorption and desorption of impurities in the feed gas.
Based on study design assumptions, high-purity hydrogen product at a rate of 8,500 kg/h is released at the top of the adsorber vessels (typically, there are four of them). Desorbed impurities leave at the bottom of the vessel as process “tail gas”, containing 34% (v) H2. This tail gas fuels the 134 MWe (gross power output) GTCC power plant driven by two Siemens SGT-800 industrial gas turbines.
Click here to find additional articles about hydrogen (H2) fueling gas turbines.
For an in-depth look at why using “green” hydrogen to fuel gas turbines does not make sense, see “Green Hydrogen Powering Gas Turbines: a Realistic Strategy?“)





