The growing consensus of the importance of green hydrogen in meeting long-term net-zero climate targets has placed sharp focus on the urgent need to reduce the high production costs using electrolyzers, both in terms of required capital investment and the high energy cost of the electrolysis process itself.
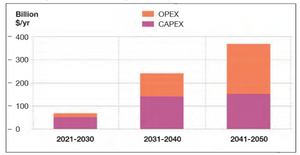
In its Hydrogen Forecast to 2050, Norway-based consultancy DNV says that green hydrogen must grow from zero today to 15% of global energy mix by mid-century to meet climate targets. They go on to say that required global expenditures (CAPEX + OPEX) to meet that goal will approach $7 trillion USD, and will require some 3,100 gigawatts of surplus renewable energy to drive the needed electrolyzer capacity.
It is also widely believed that green hydrogen represents the ultimate solution for decarbonizing gas turbine power generation and other major industrial uses currently dependent on natural gas.
Here, GTW examines the potential for meeting these goals with two commercially available electrolyzer types and a third advanced technology actively under development and demonstration:
Liquid alkaline electrolyzers. Reportedly offer the lowest $/kW capital cost for commercial scale plants (over 2 MW input), on the order of $800 to $1,000 per kW, with an efficiency of around 55 kWh input per kg of H2 product.
PEM electrolyzers. Polymer-electrolyte or proton-exchange membrane units have a higher capital cost range of $1,400 to $1,700 per kW due to costly catalysts, but exhibit somewhat higher efficiency closer to 52 kWh input per kg of H2 product with better load-following ability.
Solid oxide electrolyzers. Basically fuel-cells operating in reverse, utilizing waste heat from external sources to reduce primary energy input and achieve higher efficiencies approaching 40 kWh input per kg of H2 product.

Industry focus on high cost
According to industry reports, electrolyzer manufacturers are in general agreement on the potential for rapidly reducing capital costs through economies of scale. Many are embracing development of larger electrolyzer units while others are betting on quantity over unit size.
Regardless of approach, suppliers are already achieving significant reductions in capital expenditure (CAPEX) supported by the evolving market demand for larger systems and innovation in system design and manufacturing methods.
One example is thyssenkrupp Industrial Solutions (Germany), in partnership with De Nora (Italy), who recently announced a significant expansion of manufacturing capacity to meet growing global demand.
As Christopher Noeres, head of energy storage and hydrogen business at thyseenkrupp, told Chemical Engineering magazine: “With the expansion we can build electrolysis plants with an annual capacity of 1 gigawatt (power input), and we plan to expand even further.” (Note: 1GW of capacity equals about 160,000 metric tons of green hydrogen production per year.)
Thyssenkrupp electrolyzers are built as prefabricated skid-mounted modules which can be combined into multiple, large assemblies so hydrogen production capacity can be customized to suit site requirements.
“Scaling up production capacity,” says Noeres, “will help realize economically promising value chains. Not only for large-scale production of green hydrogen, but also for downstream uses such as the manufacture of sustainable chemicals and energy carriers such as ammonia and methanol.”
For future hydrogen economy
Electrolyzers 10 to 100 times the size of today’s largest units are envisioned in a future hydrogen economy that will effectively utilize large amounts of cheap electricity generated by high-volume wind and nuclear power.
Besides economies of scale and improved manufacturing techniques, the industry is increasing its development efforts to reduce energy consumption now typically on the order of 55 kWh per kg of hydrogen product.
Research and development is aimed at decreasing the use of precious metals, like platinum and iridium as catalysts for PEM electrolyzers, and making advancements in electrode technology for alkaline systems. (Alkaline electrolyzers typically use nickel-based catalysts.)
A major goal is to achieve “fossil parity” and produce green hydrogen for the same price as steam methane reforming (SMR) of natural gas and carbon capture for producing blue hydrogen. Today, the cost of producing green hydrogen from wind power is around 2-3 times greater than that of blue hydrogen.
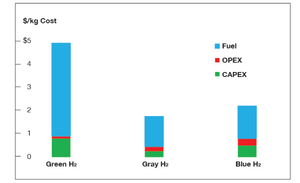
The European Commission estimates that hydrogen derived from fossil fuels costs about $2 per kg, even at current inflated natural gas prices. For comparison, cost estimates for green hydrogen range between $4.00 and $6.00 per kg, depending on cost of input electricity.
In this regard, some recent government and industry forecasts predict that the cost of renewable energy for electrolysis will fall sharply over the next decade, while global natural gas prices will continue to escalate with greater demand for LNG.
Since the difference in “fuel” cost (chart) is the main determinant of the cost gap, this will go a long way to reduce the difference in cost between green and blue hydrogen in the short term.
At the same time, it is expected that the capital cost of large-scale electrolyzer installations will fall well below today’s levels (some say by as much as 50% or more), and that electrolyzer utilization factors will continue to increase, thereby reducing CAPEX contribution to cost per unit of production. On this basis, concludes McKinsey & Co. in its Hydrogen Council report H2 Insights 2021, renewable hydrogen could break even with gray H2 before 2030, much sooner than previously expected.
To make green hydrogen competitive at scale, electrolyzer manufacturers say, they face a formidable challenge to reduce both capital (CAPEX) and operating (OPEX) costs.
Electrolysis 101
A simplified definition of electrolysis of water is the use of electricity to split water into hydrogen and oxygen molecules. This is generally recognized as the most promising pathway for carbon-free hydrogen production from renewable or nuclear energy sources.
Electrolysis takes place in a modular unit called an electrolyzer, to which electrical current is supplied through electrodes. The output of value, essentially pure hydrogen gas, is then compressed to high pressure for transport by pipeline or tube truck. (Liquefaction of hydrogen requires temperatures below -253ºC (-423ºF) or approaching absolute zero is considered impractical).
Oxygen produced by electrolysis is considered a byproduct which can be released to the atmosphere or captured for various industrial or medical uses. Although more complicated than with other industrial gases, compressed hydrogen can be stored for a relatively long period and is seen as a way of storing surplus renewable and nuclear energy otherwise curtailed.
Electrolyzers range in size from small, appliance-type equipment well suited for small scale local hydrogen production and use, to large-scale production facilities, made up of multiple trailer-size skid assemblies of “cell stacks” that could directly supply a major user or act as the base of a distribution “hub” for a cluster of users.
Due to the strong natural bond between hydrogen and oxygen in a water molecule electrolysis is unavoidably an energy intensive process. The theoretical minimum energy (primarily electrical) needed to split water electrochemical-ly is 285.8 kJ/mole of water.
This translates to just under 40 kWh per kg of H2, or a “conversion efficiency” of 83% (see curve). In the real world, the typical commercial electrolyzer requires an energy input of around 55 kWh per kg H2, for a conversion efficiency of about 60%.
In their forecast DNV says that the predicted growth in green hydrogen production by 2050 will require a surplus of renewable energy to power an electrolyzer capacity of 3,100 gigawatts. “More than double the total installed generation capacity of solar and wind today,” says DNV.
Commercial electrolyzer designs
In its most basic form, an electrolyzer contains a cathode (negative charge),an anode (positive charge), and an electrolyte. The entire system also includes pumps, vents, storage tanks, DC power supply, dryer separator, and other components. Customarily, the hydrogen compressor is considered outside of the electrolyzer system scope.
There are two main electrolyzer designs on the market today, with varying cost and performance, advantages and disadvantages. These are called alkaline, and polymer-electrolyte (aka proton-exchange ) membrane or simply PEM electrolyzers.
Another technology, evolving out of solid oxide fuel-cell technology is the solid oxide electrolyzer-cell (SOEC) which is at an advanced level of demonstration and expected to be available in the market in the foreseeable future.
Still further out in time is the anion-exchange membrane (AEM) electrolyzer, which is similar to PEM but holds promise of some important advantages and cost reductions.
Alkaline electrolyzers
The most established alkaline electrolyzer technology, also considered the most affordable, uses a caustic liquid electrolyte, usually a 25-30% aqueous solution of potassium hydroxide (KOH).
The electrodes comprising the cell stack are immersed in the liquid electrolyte, separated by a selective separator diaphragm that allows transport only of negatively charged hydroxide ions (OH-) from cathode to anode (see illustration).

Under these conditions, and helped by catalyst coating on the electrodes, hydrogen is generated at the cathode and oxygen at the anode. The separator diaphragm in the past was asbestos, but today typically made of Zirfon PERL, a proprietary membrane material developed by Agfa.
Alkaline electrolyzers, which have been commercially around since the 1950s, are available at high capacities and show good performance if operated continuously. Operation is at relatively low temperatures (60-90°C) and pressures. More recent developments allowing operation at higher pressure have improved performance and operating flexibility.
Although long considered the mainstay of the industry, liquid alkaline electrolyzers suffer from several shortcomings, particularly when looking for rapid response to changes such as found with renewable power supplies.
These issues were largely addressed by introducing the PEM electrolyzer as described below.
PEM electrolyzers
The proton-exchange or polymer electrolyte membrane (PEM) electrolyzer was introduced in the 1960s (by GE) and commercialized in the 1970s and 1980s to overcome drawbacks of liquid alkaline electrolyzers, namely poor transient and partial load performance, long startup time, low current density, and relatively low pressure operation (~30 bar max.).
In PEM electrolysis the reaction takes place in a thin cell equipped with a solid polymer electrolyte sandwiched between the electrodes.
The membrane, coated with a precious metal catalyst, replaces the liquid electrolyte, and serves multiple functions: as a proton exchange membrane, as the insulator between the electrodes, and as an effective separator between product gases.

As shown in the illustration, feed water is supplied at the anode where it reacts to form oxygen molecules (O2) and free electrons (e-), which are fed to an external circuit. The reaction also produces positively charged hydrogen ions, or protons (H+).
These migrate through the membrane and pass to the cathode where they combine with the free electrons (supplied by the external circuit) to form hydrogen gas (H2). The temperature of the PEM cell is maintained at 50-80˚C.
The efficiency of PEM electrolysis is considered comparable to that of liquid alkaline electrolysis (plus or minus).
Significant advantages of PEM electrolysis include the ability to operate at higher current densities (and, therefore, higher product throughput) and to react quickly to fluctuations in power supply or product demand.
These advantages help reduce investment and operating costs, especially for systems coupled with fluctuating and intermittent energy sources such as wind and solar.
The PEM membrane
One of the most common and commercially available PEM membrane materials is a fluorocarbon based polymer knows as Nafion, a DuPont product. While the membrane is typically thin (~100-200 μm) it allows the electrolyzer to operate at much higher internal pressures than alkaline electrolyzers (up to 350 vs ~30 bar).
So the thin membrane not only reduces the power losses associated with the conduction of protons (across the membrane) it enables a pressurized hydrogen product output, thus reducing, or even eliminating, compression requirements for storage and transport.
The PEM membrane, due to its semi-permeable structure, exhibits a low gas crossover rate resulting in high product gas purity. (Hydrogen produced by liquid alkaline hydrolysis requires stripping of caustic mist and washing.) A disadvantage of PEM is that it requires use of costly precious metal catalysts, such as platinum (on the cathode side) and iridium (on the anode side). A primary goal of R&D efforts is to reduce the catalyst “loading” or to eliminate the need for catalyst altogether.
In that regard, a newer variant of membrane-type electrolyzer, still in development, the anion-exchange membrane (AEM) electrolyzer, avoids the use of the costly precious metals. Like alkaline electrolysis, AEM allows negatively charged ions (OH-) to pass through the membrane. The process is also more effective at smaller-scale, making it suitable for decentralized applications.
Solid oxide electrolyzers
Solid oxide electrolyzer cells (SOEC) are essentially solid oxide fuel cells operating in reverse or regenerative mode to achieve electrolysis of water. Like fuel cells, the most common SOEC electrolyte is a dense solid ionic conductor consisting of zirconium oxide (ZrO2) doped with yttrium oxide (Y2O3).
When a voltage is applied across the electrodes, water (fed as superheated steam into the porous cathode) is reduced to form hydrogen gas (H2) and negatively charged oxygen ions (O2-). The latter selectively pass through the ceramic conductor and react at the anode to form oxygen gas (O2).
Because of its high melting temperature ZrO2 enables operation at higher temperature than either alkaline or PEM electrolyzers. For the ion conductor to operate properly, SOEC units operate at temperatures on the order of 700-800°C (~1300-1400°F).

This offers the potential for much lower electrical energy consumption by effectively using waste heat available from external sources to supply some of the energy needed for electrolysis. (However, this need for an external heat source may limit application of SOEC somewhat.)
So, according to recent reports (e.g., see PV magazine, March 2021), instead of requiring ~55 kWh of electrical energy to produce 1kg of hydrogen, as is typical of alkaline and PEM electrolyzers, high temperature SOEC electrolyzers will require on the order of 40 kWh per kg H2.
This greater than 25% reduction in electrical energy input per kg roughly translates to an increase in electrolyzer “energy efficiency” from 60% to 80% based on H2 energy output divided by electrical energy input.
Emerging R&D into alternative solid oxide electrolyzers based on proton-conducting ceramic electrolytes shows promise for lowering the operating temperature somewhat to 500-600°C (about 900-1100°F).
Comparing electrolyzer technologies
Electrolyzers are commonly rated in terms of kW or MW input power capacity, and capital cost is expressed in terms of $ per kW.
Alkaline electrolyzers are generally considered to have the lowest cost per kW in commercial scale plants (over 2 MW), on the order of $800 to $1000 per kW– which works out to $1.8 to $2.25 million USD per ton of daily capacity (based on 55kWh per kg hydrogen production).
PEM electrolyzers weigh in at an overall higher capital cost of $1,400 to $1,700 per kW (or $3.3 to $4.2 million USD per ton of daily capacity).
The price difference between alkaline and PEM electrolyzers is largely explained by relatively immaturity of PEM technology and use of precious metals in PEM electrolyzers.
There is much uncertainty regarding likely investment cost of SOEC due to pre-commercial status of the technology. But the 25% reduction in input power required and other advantages of high temperature and high-pressure operation make it economically competitive.
Need for R&D to cut cost
According to the US DOE, today’s production cost of more than $5 per kg for green hydrogen (from wind power) is more than double the cost of blue hydrogen produced from natural gas using conventional steam-methane reforming (SMR) technology with carbon capture.
With the ultimate stretch goal set at $1/kg (perhaps more realistically at $2/kg) the US and other international government agencies and industry groups have launched major efforts to cut cost:
The DOE has outlined an R&D plan, identifying the main challenges related to electrolyzer systems as:
1) better understanding of performance, cost and durability trade-offs,
2) reducing capital cost,
3) improving energy efficiency, and
4) increasing operational life.
Where the process depends on use of precious metal (e.g., platinum, iridium) catalysts, the reduction of catalyst loading is one of the important routes toward reducing capital cost. So too would be development of less costly alternative catalysts.
Cost reduction may also be realized through economies of scale, including increased unit size and mass production of cell stacks, and simplification of balance of plant systems. And by engineering improvements such as more conductive membranes and higher process pressurization operation for direct hydrogen storage.
But comparative evaluations involve trade-offs. For instance, low current density may reduce power usage, but also increase capital cost per unit of product and negate any gains in efficiency.
Read this (provocative) counter-point: Green hydrogen powering gas turbines: a realistic strategy?





