At the time of writing, after nearly a decade of media blitz and hype, even laypeople are readily conversant in colors of hydrogen and what wonders they can do in decarbonizing global economies including electric power generation, transportation, and the industrial sector (e.g., cement and steel-making).
The focus of this article is exclusively on utility or grid-scale electric power generation with gas turbines burning hydrogen. Other uses of hydrogen of any “color”, including existing and/or projected, e.g., fuel cells for transportation and distributed power generation, at different scales and concomitant infrastructure needs, are outside the scope of the article.
Also this discussion is strictly quantitative based on publicly available information and simplified calculations using first principles without resorting to starry-eyed “opinions” or “prognostications”.
How hydrogen (H2) can help decarbonization in technologies involving combustion is obvious: the product gas does not contain CO2 (except the minuscule amount that came in with ambient air). This, of course, is wonderful news. The bad news is that, unlike natural gas (mostly methane), H2 is not naturally available for extraction and utilization; it has to be manufactured first.
The worse news is that manufacturing H2 is extremely expensive, not only in terms of dollars and cents but also in terms of kilowatts. Finally, to make matters truly worse, presently, about 95% of H2 production in the U.S. (globally, more like 75%) is via steam methane reforming (SMR), which is a very complex thermo-chemical process and, while producing the sought-after hydrogen, generates a significant amount of CO2 emissions.
This article looks at the various processes used to manufacture hydrogen by referring to the “color” of hydrogen associated with each process, and offers an analysis of hydrogen strategies that further the goal of decarbonization.
Green Hydrogen
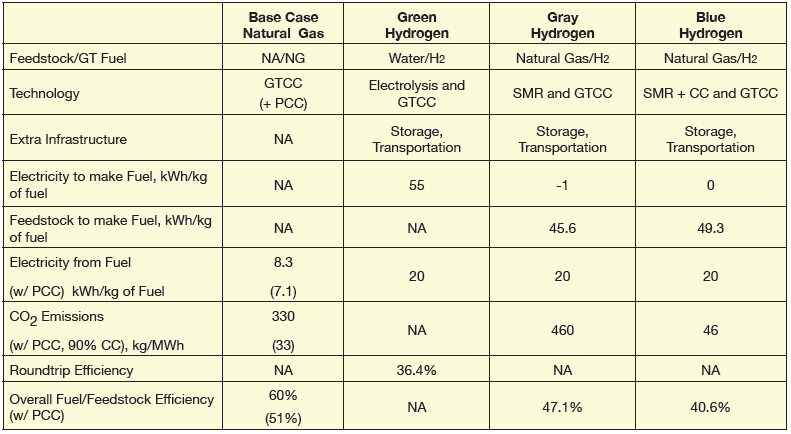
This is where electrolysis of water comes into play. It is a “green” technology because no CO2 is produced in the process. The underlying chemical reaction is very simple but consumes a lot of energy, which is supplied by a battery (in the laboratory) or by a power plant (in a commercial-scale application).
It can be shown that the minimum theoretical energy requisite for the electrolysis of water to take place is 237.1 kJ/mol of H2O, which translates into approximately 33 kWh/kg of H2. This is the yardstick that one must use to gauge the claims made by electrolyzer manufacturers.
For example, Proton exchange membrane (PEM) electrolyzers are commercially available today and are rapidly gaining market traction as, among other factors, they are more flexible and tend to have a smaller footprint compared to their forerunner technology, the liquid alkaline electrolyzer .
Published numbers for PEM and other electrolysis technologies cover a broad range and, with little or no commercial-scale experience, verifying them is quite difficult. A conservative number for state-of-the-art PEM electrolyzers is 55 kWh/kg, which will be used herein.
Future developments can (maybe) bring it down to 45 kWh/kg. Interested readers can easily modify the findings below using the number that they think is “correct”. Rest assured that the proverbial needle will not move significantly.
The objective in green hydrogen production is to utilize renewable resources, e.g., solar or wind, to run large electrolyzers to make H2, and then store, transport, and burn it in gas turbine combustors. Easier said than done.
Let us consider an advanced class 60-Hz gas turbine rated at 400 MWe and 43% net LHV efficiency, with 400 / 0.43 = 930 MWth of heat (fuel energy) consumption (see Figure 1). In other words, ignoring small changes in efficiency when changing fuel from natural gas to hydrogen, this gas turbine with a modern Dry-Low-NOx (DLN) combustor, if it could handle 100% H2 fuel (not possible presently), would consume 930 / 120 = 7.75 kg/s of H2 (with 120 MJ/kg LHV).
To produce H2 at this rate in an electrolyzer, one would need 55 kWh/kg x 3,600 s/h x 7.75 kg/s = 1,535,000 kWe, i.e., 1,535 MWe or more than 1.5 GWe, electric power. That’s nearly four times the electric power produced by the hydrogen-burning gas turbine itself and amounts to a roundtrip efficiency (RTE) of about 26%.

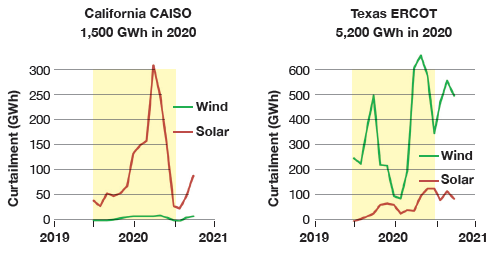
According to the 2021 Land-Based Wind Market Report of U.S. DOE’s Office of Energy Efficiency & Renewable Energy, selected 2020 statistics were as follows: 122 GW of installed wind capacity, 36% fleet-wide capacity factor, and 3.4% curtailment across all U.S. Independent System Operators (ISOs), with 5% in Midcontinent ISO (MISO) and 4.6% in ERCOT, Texas. Thus, in 2020, total wind curtailment can be calculated as 122 GW x 8,760 h x 36% x 3.4% = 13,000 GWh.
Consequently, if the entire curtailed wind generation in the U.S. was dedicated to H2 production via electrolysis, it would sustain only two (2) advanced class gas turbines with 4,000 hours of annual operation, i.e., 13,000 GWh / (1.5 GWh per GT x 4,000 h) = 2.17 GTs.
If these two gas turbines were operating in combined cycle (GTCC) with 63% net LHV efficiency, their output would be 586 MWe each. Thus, 13,000 GWh of power expenditure would support 5,000 GWh of electricity production for an RTE of 5 / 13 = 38.5% (ignoring the energy consumed to store and transport H2 plus leaks, losses, and miscellaneous inefficiencies).
In the calculation above, ISO base load rating numbers were used. According to 2020 (final) EIA Form 923 data, the most efficient GTCC power plant in the U.S. generated 9.5 million MWh at an effective LHV efficiency of 59.4%. If this power plant could repeat the same performance with 100% H2 fuel, it would consume 478,300 metric tons of H2, which would require 26,300 GWh of power to produce via electrolysis (corresponding to 36% RTE – again, ignoring everything between the electrolyzer and the gas turbine combustor).
As calculated above, in 2020, only half of this amount of “surplus” GWh were available via the entire U.S. curtailed wind generation. Let us assume that this plant burned a blend of H2 (equal to the amount that can be generated with 13,000 GWh of – maximum possible in 2020 – curtailed wind generation) and methane (a proxy for natural gas) for the same exact, field-recorded performance. This would lead to a reduction in CO2 stack emissions by about 1.6 million metric tons, bringing the specific plant CO2 emissions down to about 168 kg/MWh (370 lb/MWh) from 334 kg/MWh (735 lb/MWh).
According to the U.S. EPA, in 2020, U.S. greenhouse gas (GHG) emissions totaled 5,222 million metric tons of CO2 equivalent after accounting for sequestration from the land sector. (Electricity production accounted for 25% of that amount.) Thus, the reduction that could have been achieved by using the entire U.S. wind curtailment for H2 production, to be used in the most efficient GTCC power plant in the country, would result in a reduction of 1.6 / 5,222 ~ 0.03% in total GHG emissions.
As a yardstick, consider that, per U.S. EPA, GHG emissions decreased from 2019 to 2020 by 11%. The primary major driver for the decrease was attributed to reduced CO2 emissions from fossil fuel combustion, primarily by replacing coal-fired generation with natural gas.
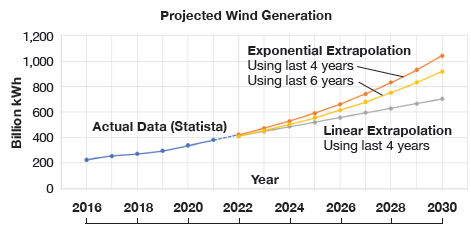
Extrapolation of the growth trend in wind generation of the last four years suggests that, by 2030, expected generation will be between 700 (linear growth) and 1,000 (exponential growth) billion kWh (see Figure 3). Assuming 5% curtailment rate, this corresponds to 35,000 to 50,000 GWh of curtailed wind generation.
Going with the higher number, assuming 50 kWh/kg electrolyzer technology and 60% annual average (field performance) GTCC efficiency, 20 million MWh of electricity could be generated with 100% H2 fuel. This would save about 6.5 million metric tons in CO2 stack emissions, which is equivalent to 6.5 / 5,222 ~ 0.13% of 2020 total U.S. GHG emissions (or 0.52% of GHG emissions from electricity production).
Gray Hydrogen
Let us now turn to manufacturing hydrogen via SMR, a more-than-a-century old technology utilized around the world to make H2 to be used in the chemical process (for production of ammonia) and refining (for hydrocracking and desulfurization of fuels) industries.
From the chemical reaction formulae, on a theoretical basis, each kilogram of H2 produced comes with 5.5 kg of CO2 production. In the actual SMR plant (see simplified flow diagram, Figure 4), one must include the fuel burned in the reformer as well. This can raise the CO2/H2 ratio to about 9.2 (it can be as high as 10 or 11).
Let us apply this to our sample gas turbine (400 MW at 43% LHV) burning 7.75 kg/s H2. The SMR process supplying the requisite amount of hydrogen would generate 257,000 kg/h of CO2. On a simple cycle basis, this corresponds to about 642 kg/MWh (1,415 lb/MWh) CO2 generation. On a combined cycle basis, the number is 438 kg/MWh (966 lb/MWh).
In comparison, for the best field-performer natural gas fired GTCC of 2020 cited earlier in the article, with ~60% LHV efficiency instead of 63%, which is the basis of the SMR numbers cited above, the corresponding number is 333 kg/MWh (735 lb/MWh), or more than 30% less than the one fired on 100% gray hydrogen.
The author is confident that the readers will agree that no further discussion is necessary; “gray” hydrogen is a non-starter.

Blue Hydrogen (SMR + CCS)
Of course, the SMR plant can be retrofitted with carbon capture and sequestration (CCS) to produce “blue” hydrogen.
In an SMR plant, CO2 could be captured at three locations: (i) from the pressure swing adsorption (PSA) unit’s tail gas, (ii) from the reformer flue gas with about 90% efficiency (~45% (v) and ~20% (v) concentration, respectively and less than one bar partial pressure), or (iii) from the raw H2 at the shift reactor exit with 99+% efficiency (~15% (v) concentration and ~3.5 bar partial pressure).
The removal technologies available include amine-based scrubbing, physical solvents and membranes and there is widespread experience in chemical process industry in CO2 removal from raw H2 at high pressure. In contrast, CO2 scrubbing from the GTCC flue gas at low partial pressures and high volume flows requires larger and more expensive equipment and consumes more parasitic power. In any event, from a purely CO2 emissions perspective, if the SMR plant is fitted with 90% CCS, for our example GTCC firing “blue” hydrogen produced in that plant would have 44 kg/MWh (97 lb/MWh) CO2 emissions (ignoring everything between the SMR hydrogen delivery point and the gas turbine combustor).
The last number is quite attractive, however, there is absolutely no reason why our “best in class” (based on field performance) GTCC cannot itself be retrofitted with a 90% post-combustion carbon capture (PCC) system as well.
In that case, its CO2 stack emissions would drop to 33 kg/MWh (74 lb/MWh), almost 30% better than blue hydrogen case with SMR plus CCS. (Note that, CO2 production of the SMR generating the requisite H2 for this plant would be 465 kg/MWh or 46.5 kg/MWh with 90% capture.)
In other words, unless a compelling CAPEX/OPEX argument can be made in favor of blue hydrogen (via SMR plus CCS), it is impossible to see how it can be a viable alternative to natural gas-fired GTCC with PCC. Even then, to bring the latter to a par with blue hydrogen in terms of kg of CO2 per MWh of generation (assuming 90% capture), 86% capture is sufficient with concomitant savings in PCC block size and CAPEX/OPEX.
Considering the first-of-a-kind nature of the final systems with CCS (not SMR or GTCC on their own) as well as the uncertainty and “unknown unknowns” in key infrastructure items such as H2 storage and transportation (including pipeline construction and parasitic power of compression) and CO2 sequestration and/or utilization, any attempt to make economic comparisons without bona fide front-end engineering and design (FEED) studies done by credible EPC contractors involving major OEMs (a multi-year, multi-million dollar effort) is a futile endeavor. (The reader can consult the DOE-funded General Electric and Southern Company FEED for retrofitting a GTCC with PCC at Plant Barry – Alabama Power. Results are expected in early 2023. Also see page 16 of 2022 GTW Handbook.)
In passing, it should be mentioned that there are other technologies to manufacture blue hydrogen from natural gas feedstock. Examples are partial oxidation (POX) and autothermal reforming (ATR). The latter is an oxy-combustion process combining POX and SMR. The main advantages of ATR vis-à-vis SMR are the increased energy efficiency, faster start-up times, and faster response times to transient events. Developed in 1950s, ATR is used to generate syngas for ammonia and methanol synthesis.
A key drawback of ATR is that it requires an air separation unit (ASU) to make the oxygen for combustion. The key attractive feature of ATR is that it generates 20% less CO2 to generate the same amount of H2 as SMR. The reader can find many published references comparing SMR, ATR, and POX as well as their variants in terms of cost and performance. Since none of those technologies competing with the time-tested SMR for blue hydrogen is even close to large-scale commercial application, a closer look at them in this short article is not warranted.
Blue Hydrogen (Gasification + CCS)
Globally, about 75% of H2 production is from SMR and the rest mainly from gasification. In gasification process, the first step is reacting coal (or another liquid/solid hydrocarbon feedstock) with oxygen and steam under high pressures and temperatures to form synthesis gas (syngas), a mixture consisting primarily of carbon monoxide (CO) and hydrogen.
After the impurities are removed from the syngas, the CO in the gas mixture is reacted with steam through the water-gas shift reaction to produce additional H2 and CO2. Hydrogen is removed by a separation system, and the highly concentrated CO2 stream can subsequently be captured and stored.
In published studies, CAPEX for this technology is estimated at three times that for SMR with CCS. As emphasized earlier, without a comprehensive FEED study involving both technologies, the author would refrain from commenting on such findings. The “true” ratio can be somewhat less or significantly larger. Suffice to say that the economic feasibility of this route to hydrogen production is rather iffy.
But, before writing gasification off, let us pause here for a moment. Steam-methane reforming uses natural gas as feedstock to manufacture H2. Yet, natural gas is the “cleanest” fossil fuel that can be used directly for electric power generation in the most-efficient combustion-based technology today, namely, advanced class GTCC.
Under the light of the calculations presented above, it is a mystery to this author, why anyone would go to the trouble of first converting natural gas to H2 with CCS (which can be appended directly to the GTCC in the first place) and piling complex process upon complex process (some nonexistent and extremely costly to build and operate) to then burn the hydrogen in the same GTCC.
However, there is a rationale behind making blue hydrogen via gasification of coal, refinery residue, or other problematic hydrocarbons. Unlike the electrolysis or SMR (or its derivatives or close relatives), in this case, the feedstock, in contrast to surplus electric power plus water or natural gas (mostly methane, that is), is typically not considered to be a good candidate for ‘clean’ electric power generation.
As an example, let us consider visbreaker residue, which is pretty nasty stuff (its LHV is about 39 MJ/kg). Under normal circumstances, this residual heavy oil is not suitable as a gas turbine fuel. Still, with a GTCC based on an E or (maybe) F class gas turbine equipped with diffusion flame combustors with steam injection, fuel preparation with washing and additives, and a two-pressure, non-reheat HRSG with high stack temperature (to prevent sulfuric acid condensation on economizer tubes), it can be used to generate electric power with nearly 600 kg/MWh CO2 stack emissions (not to mention SOx and other toxic stuff).
In contrast, utilizing the same feedstock in a gasification plant comprising a POX reactor (utilizing O2 generated in an ASU), water-gas shift reactor, a Selexol process to separate H2S and CO2 from the syngas, and a PSA block to generate “blue” hydrogen for utilization as fuel in an advanced class GTCC is quite attractive (ignoring CAPEX and OPEX, of course).
Electric power generated this way is calculated to have about 60 kg/MWh CO2 stack emissions but no SOx. Specific electricity output is about 13.1 MJe per kg of feedstock. In comparison, for the residual oil-burning GTCC example above, with no capture, specific electricity output is 18.8 MJe/kg. With capture and extra flue gas cleanup, it can easily drop to 15 MJe/kg.
Clearly, it is impossible to make a judgement call with such “back of the envelope” calculations. It is, however, quite obvious that, if the CAPEX and OPEX numbers can be made to work out, gasification of a “not green” feedstock such as coal, petcoke, refinery residues, etc. (or biomass for that matter) with CCS to produce blue H2 can be a viable way to clean, sustainable electric power generation while making use of a wider range of readily available and otherwise low-value resources.
Recap
What green hydrogen from renewable-powered electrolysis can exactly accomplish in terms of reducing CO2 emissions from grid-scale electric power generation is not clear at all to the author. Even using the entire curtailed wind generation in the U.S. to make H2 and burn it in GTCC cannot make a proverbial pin-prick in GHG emissions.
Using the best possible assumptions, including 100% H2 burning DLN combustors, the impact is much smaller than 1% – not surprising because the generated H2 can only support a couple of state-of-the-art GTCC power plants at most. One can certainly throw in curtailed solar energy into the mix, but this will not change the answer by a whole lot. (Consider that, per U.S. EIA, in 2020, CAISO curtailed 1.5 million MWh of utility-scale solar (vis-à-vis 13 million MWh curtailed wind for the entire U.S.), which is 5% of total production.)

In passing, advanced gas turbine DLN combustors can only handle 30% (by volume) H2 in fuel gas (although one major OEM claims the ability to use 50%(v) H2 for its latest advanced DLN combustor design). This achieves only 10% reduction in CO2 emissions on kg/MWh basis (see Figure 5). For 50% reduction, H2 in fuel gas should be 75%(v).
It should be pointed out that gas turbines equipped with conventional, diffusion flame combustors and H2O injection for NOx control can and do burn 100%(v) H2 today (in refinery and steel mill syngas applications). Their water consumption is offered as a disadvantage while keeping quiet about water consumed in electrolysis. Readers can draw their own conclusions as to why “thought leaders” are silent about this option, which is ready to be deployed today.
Finally, per U.S. EIA (June 9, 2021), over the past 15 years, the U.S. electricity generation mix has shifted away from coal and toward natural gas and renewables, resulting in lower CO2 emissions from electricity generation. In 2019, the U.S. electric power sector produced 1,724 million metric tons of CO2, 32% less than the 2,544 million metric tons produced in 2005. Now, this is real decarbonization!
The basic premise of gray hydrogen is that one takes a perfectly “clean” (relatively speaking) feedstock like natural gas, which you can burn in a state-of-the-art GTCC at almost 60% field-measured efficiency, use it to make H2 while pumping extra CO2 into the atmosphere, and then burn it in the same GTCC. One can certainly think of a scenario where the product of an SMR facility, whose existing customers have disappeared, can be kept running and making H2 to be used as gas turbine fuel. Even then, one might wonder whether it is not better to retire the facility in the first place to help lower GHG emissions. Alternatively, and this may be even a better proposition, such facilities can be retrofitted with CCS to provide blue hydrogen for use in industrial and chemical process facilities. (Energy storage is another possibility but unlikely because such industrial SMR plants tend to run round the clock.)
Blue hydrogen eliminates the CO2 disadvantage of gray hydrogen by throwing in a CCS facility into the mix. But the basic question still remains. A GTCC equipped (or retrofitted) with PCC can burn natural gas directly and still do better in terms of bottom line CO2 emissions. There are some arguments, primarily based on the ease of CO2 capture (vis-à-vis PCC applied to GTCC flue gas) and its lower cost, but without a serious FEED study, the numbers, quite close to begin with, have to be taken with a grain of salt. The basic premise is as convoluted as pointing to your left ear with your right hand while putting your right arm behind your head, instead of just using your left hand and simply raising it.
Blue hydrogen from partial oxidation (POX) or gasification of coal and a variety of “nasty” feedstocks, on the other hand, is a perfectly rational proposition. It enables us to make use of an otherwise undesirable but readily and cheaply available feedstock such as coal and other waste products of chemical/refinery industries in grid-scale electricity generation. In the end, it is highly likely that it will not turn out to be economically feasible. Nevertheless, the requisite technology is available and deserves a close look.
In fact, readers can read about a three-phase demonstration project in Japan (Osaki CoolGen Project), which recently demonstrated the operation of an oxygen-blown, coal-fired integrated gasification combined cycle. In the second phase, which is currently ongoing, the designers of the facility plan to demonstrate 90% CO2 capture (99% purity) from the syngas via physical absorption (using Dow’s Selexol Max solvent). Scrubbed syngas contains about 85% (by volume) H2 and used as a fuel for the gas turbine. In the third phase of the project, the objective is to utilize H2 in a fuel cell to produce power.
As a final note, it should be emphasized that the concept of hydrogen economy is nothing new. It popped onto the scene half a century ago (e.g., see Chapter 18, p. 117 of “Energy and the Future” by Hammond, Metz, and Maugh II, AAAS, 1973). There is no doubt that there is a spot for hydrogen in a comprehensive technology portfolio for sustainable energy. As more and more renewable resources are deployed, on a case by case basis, increased use of green hydrogen as a storage medium and gas turbine fuel will be a reality as well.
However, unless the world transitions to a fully “distributed generation” mode, the expectation that it will be the proverbial knight in shining armor for zero-carbon electricity, especially with 500+ MWe super-heavy gas turbines burning exorbitant amounts of manufactured green hydrogen fuel, is not realistic.
Author’s Note: This article is largely based on recent work done by the author, e.g., a presentation made in May at POWERGEN 2022 in Dallas, TX; a paper presented in June at ASME Turbo Expo 2022 in Rotterdam, The Netherlands; an article in Gas Turbine World, September 2021; and several chapters in an upcoming book that will be published by the Cambridge University Press (Gas & Steam Turbine Power Plants – Applications in Sustainable Power) that should be available by the end of this year.





